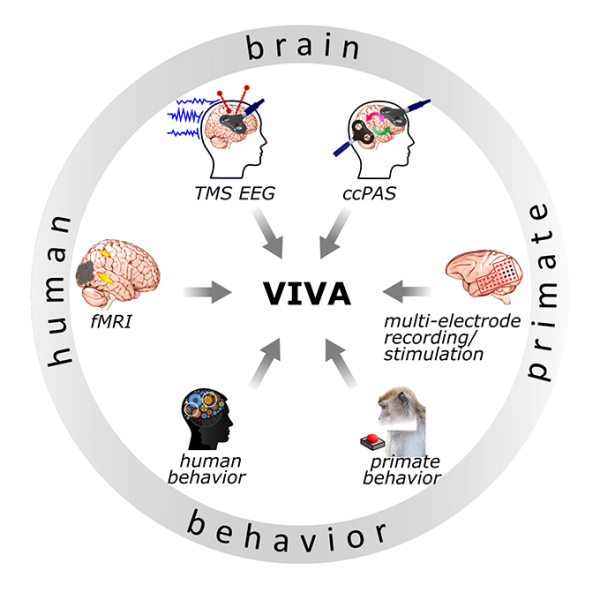
Il nostro gruppo sviluppa, sfrutta e combina tecniche comportamentali e neurofisiologiche all’avanguardia per uno studio etologicamente rilevante del cervello dei primati, in condizioni più naturalistiche possibile.
La comprensione del cervello durante il comportamento naturale è fondamentale per ottenere informazioni ecologicamente valide sulle funzioni dei circuiti neurali e sulle relative alterazioni, alla base di malattie neurologiche e neuropsichiatriche.
Per raggiungere questi obiettivi, impieghiamo tecniche all’avanguardia per la registrazione simultanea di più neuroni con tecnologie wireless, monitorando al contempo il comportamento libero degli animali in contesti individuali e sociali con sistemi multi-camera.